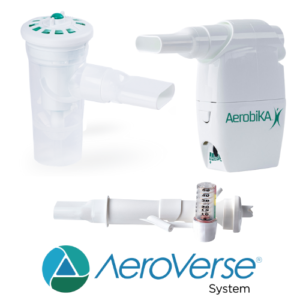
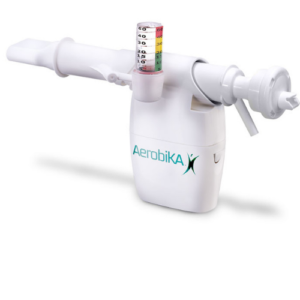
AEROBIKA® OPEP Device with Manometer Adapter
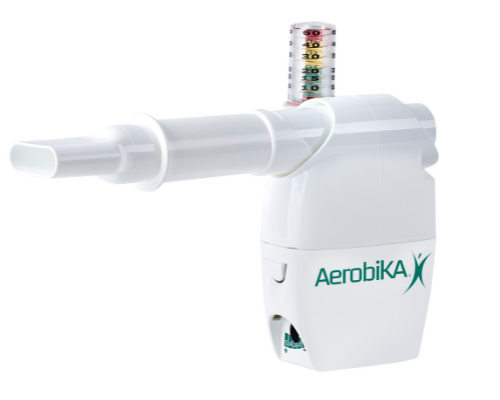
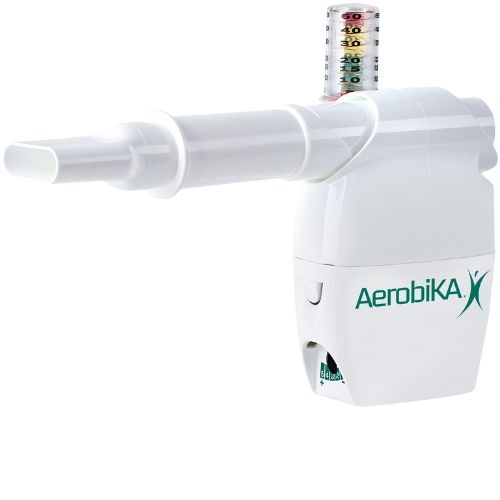
The AEROBIKA® Oscillating Positive Expiratory Pressure (OPEP) Therapy System with the manometer adapter helps monitor expiratory pressures while providing visual feedback on exhalation.
As the user exhales, the manometer gauge will rise. For each exhalation effort, the goal is to keep the gauge within the green zone (ideally between 10-20 cm H20) to maximize treatment effectiveness. Use of the manometer adapter may help patients unfamiliar with the proper breathing technique for OPEP therapy but is also a helpful reminder for seasoned users.
The AEROBIKA® OPEP device is convenient, user-friendly and provides clinicians and their patients with a means for reliable, drug-free, natural airway clearance. It opens weak or collapsed airways by creating positive pressure and oscillations simultaneously, which helps move mucus to the upper airways where it can be coughed out.
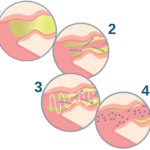
- Before Use: Airways inflamed, collapsed, and plugged with mucus.
- As you exhale through the device, positive pressure will hold the airways open. Think of how you feel when you blow up a balloon. It is the positive pressure that holds the balloon open. Inside your lungs, the positive pressure opens up small airways that may be blocked by mucus.
- As the airways open, the device switches quickly between higher and lower resistance. This may feel like vibrations or pulses which act to loosen the mucus from the walls of your lungs. The frequency of the oscillations closely match the ones within your lungs, helping move mucus to the larger airways. This makes it easier for you to cough up the secretions.
- With the airways opened and the mucus moved out of the airways, your inhaled medication may work better.2
Instructions for Use/Indications for Use
Quick Start Guide
Patient Information Leaflet
For true on-demand therapy.
Immediate feedback with a visual goal.
Purposely designed to be used together.
adding positive expiratory pressure helps
mobilize secretions and treat atelectasis.
Encourage better patient technique for both therapies.
When PAP or OPEP alone isn’t enough.
FAQ
Frequency of use will be determined by the patients’ disease state, their ability and the goal for using the device. There is no one-size fits all answer. Ideally, patients will work up to being able to use the device twice each day for 10-20 minutes each time. Clinical judgement should be used in determining what is right for each patient.
The approved cleaning methods are: soap and water or top rack dishwasher
The approved disinfection methods are: boiling for 5 minutes, 70% isopropyl alcohol soak for 5 minutes, 3% Hydrogen Peroxide soak for 30 minutes, or microwave steam bag (according to manufacturer’s instructions).
We do not recommend use with a mask. In order to maintain a good seal, an unvented mask would need to be used. These types of masks have soft, flexible edges to make it easier to prevent leaks. However, the same properties that make them ideal to create a seal may also impact the effectiveness of the treatment you are giving. The patient would still need to hold the mask and device for the duration of treatment, so we recommend use with a mouthpiece.
The recommended replacement period for the device is 12 months. If prescribed during a hospital stay, the device can be taken home by the patient for continued use. 30-day post-discharge data has shown this can reduce readmissions for COPD exacerbations and post-operative patients by 28% and 39% respectively.
If you have a question that was not answered here, please contact us.
Clinical Evidence
- A quick view of the most recent peer-reviewed evidence supporting the chambers. Learn more.
- An evidence assessment for the integration of the AEROBIKA® OPEP device into the care pathway for COVID-19 patients with airway clearance needs. Watch the video.
- 28% decrease in readmissions for COPD exacerbations. Learn more.
- 39% decrease in all-cause rehospitalizations and lower costs for post-operative recovery. Learn more.
References
- Svenningsen S. et al. COPD 2016;13(1):66 – 74.
- V Wolkove N, et al. CHEST 2002;121(3):702-7.